Plz explain ion-dipole interactions
An ion-dipole force is an attractive force that results from the electrostatic attraction between an ion and a neutral molecule that has a dipole.
- Most commonly found in solutions. Especially important for solutions of ionic compounds in polar liquids.
- A positive ion (cation) attracts the partially negative end of a neutral polar molecule.
- A negative ion (anion) attracts the partially positive end of a neutral polar molecule.
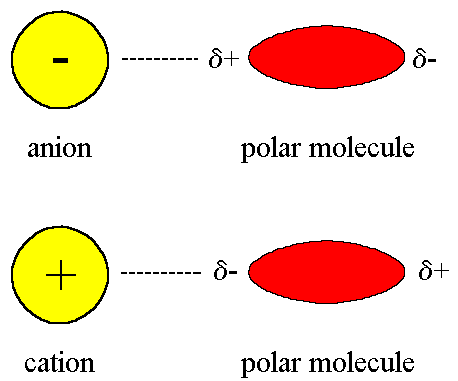
- Ion-dipole attractions become stronger as either the charge on the ion increases, or as the magnitude of the dipole of the polar molecule increases.


